Redox-responsive amphiphilic camptothecin prodrug nanoparticles for targeted liver tumor therapy†
Abstract
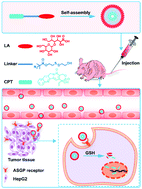
Tumor cell-targeting drug delivery systems are of great importance to anti-tumor therapy in clinics. Owing to the overexpression of the asialoglycoprotein receptor (ASGPR) on the membrane of hepatoma carcinoma cells, the conjugation of lactose on the surface of drug delivery systems has already shown significant advantages for targeting tumor cells. In this study, a disulfide bond-conjugated prodrug targeting delivery system consisting of camptothecin (CPT) and lactose (LA) was synthesized, which was denoted as CPT-S-S-LA. Camptothecin and lactose act as the chemotherapy drug and targeting ligand in the drug delivery system, respectively. Since CPT-S-S-LA is an amphiphilic compound, it can self-assemble into nanoparticles with a diameter of around 110 nm. The CPT-S-S-LA nanoparticles displayed controllable drug release behavior in the physiological environment. Unlike the free CPT, the CPT-S-S-LA nanoparticles firstly assembled at the tumor sites via the enhanced permeability and retention (EPR) effect, and then were phagocytized by the tumor cells with ASGP receptor-mediated endocytosis. Finally, the antitumor agent CPT was released for killing tumor cells, which have a high glutathione (GSH) concentration environment. The nanoparticles displayed favorable ability to target hepatoma carcinoma cells rather than the normal HUVEC cells in vitro. Both the in vitro and in vivo studies demonstrated that the CPT-S-S-LA nanoparticles display enhanced antitumor ability and reduced side effects. Thus, active targeting prodrug delivery systems should be a promising strategy for liver tumor therapy.


 Please wait while we load your content...
Please wait while we load your content...