Plasmonic gel nanocomposites for detection of high energy electrons
Abstract
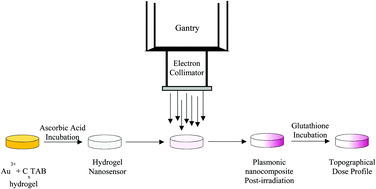
Radiation therapy is a common treatment modality employed in the treatment of cancer. High energy photons are the primary source of radiation but when administered, they leave an exit dose resulting in radiation damage to the adjacent healthy tissues. To overcome this, high energy electrons are employed in cases of skin cancer to minimize radiation induced toxicity. Despite these advances, measurement of delivered radiation remains a challenge due to limitations with existing dosimeters including labor intensive fabrication, complex read-out techniques and post-irradiation instability. To overcome these limitations, we have developed a novel colorimetric plasmonic gel nanocomposite for the detection of therapeutic levels of radiation delivered in electron beam therapy. The plasmonic nanocomposite consists of an agarose gel matrix encapsulating precursor gold ions, which are reduced to gold nanoparticles as a result of exposure to high energy electrons. The formation of gold nanoparticles renders a change in color to the agarose matrix, resulting in the formation of plasmonic gel nanocomposites. The intensity of the color formed exhibits a linear relation with the delivered electron dose, which can be quantified using absorbance spectroscopy. The plasmonic gel nanocomposites were able to detect doses employed in fractionated electron therapy, including in an anthropomorphic phantom used for planning radiation treatments in the clinic. Furthermore, the use of glutathione as a quenching agent facilitated qualitative and quantitative spatial mapping of the delivered dose. Our results indicate that the ease of fabrication, simplicity of detection and quantification using absorbance spectroscopy, determination of spatial dose profiles, and relatively low cost make the plasmonic gel nanocomposite technology attractive for detecting electron doses in the clinic.
- This article is part of the themed collection: Hybrid Nanoparticle Composites

 Please wait while we load your content...
Please wait while we load your content...