Human nail bed extracellular matrix facilitates bone regeneration via macrophage polarization mediated by the JAK2/STAT3 pathway †
Abstract
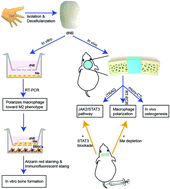
Critical-sized bone defects caused by trauma, tumor resection or serious infection represent one of the most challenging problems faced by orthopedic surgeons. However, the construction of bone grafts with good osteointegration and osteoinductivity is a clinical challenge. It has been elaborated that the nail bed tissue is an essential element for digit tip regeneration, suggesting that the nail bed may serve as a new material to manipulate bone regeneration. Herein, it was found that human nail bed extracellular matrix derived from amputated patients stimulates macrophage polarization toward a pro-healing phenotype and the expression of BMP2, to facilitate the osteogenic differentiation of bone marrow stromal cells (BMSCs) in vitro. The in vivo osteogenic capacity of decellularized nail bed scaffolds was then confirmed using a rat model of critical-sized calvarial defects. The in-depth analysis of immune responses to implanted scaffolds revealed that macrophage polarization toward the pro-regenerative M2 phenotype directs osteogenesis, as confirmed by macrophage depletion. A combination of proteomics analysis and RNA interference verified that the JAK2/STAT3 pathway is the positive regulator of macrophage polarization initiated by the decellularized nail bed during the promoted osteogenesis process. Thus, the decellularized human nail bed scaffold developed in this work is a promising biomaterial for bone regeneration.


 Please wait while we load your content...
Please wait while we load your content...