Nanoparticles with surface features of dendritic oligopeptides as potential oral drug delivery systems†
Abstract
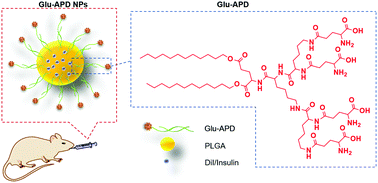
Surface features are key to the transcellular transport of nanoparticles (NPs) across intestinal epithelium cells. Endowing the NPs with specific surface features adapted to the physiological conditions of the gastrointestinal (GI) tract holds great potential for the oral delivery of peptide/protein drugs. Therefore, in this work, a glutamic acid conjugated amphiphilic dendrimer (Glu-APD) was synthesized to replace the widely used 1,2-distearoyl-sn-glycero-3-phosphatidyl-ethanolamine-polyethylene glycol (DSPE-PEG) in the preparation of poly(lactic-co-glycolic acid) (PLGA)-based NPs. Glu-APD could provide the formed NPs (Glu-APD NPs) with specific surface features of dendritic oligopeptides. With such surface features, Glu-APD NPs exhibited a 7.78-fold increase in cellular uptake and a 2.17-fold increase in the transepithelial transport amount compared with those of the DSPE-PEG2000 modified counterparts (P NPs). Instead of a dominant clathrin-mediated endocytosis as shown by P NPs, Glu-APD can provide the NPs with optional endocytosis pathways (i.e. clathrin-mediated, caveolae-mediated and micropinocytosis pathways), with the involvement of oligopeptide transporters and amino acid transporters, subsequently leading to a broadened intracellular trafficking route via the endoplasmic reticulum (ER) and Golgi apparatus. Furthermore, L-glutamic acid (L-Glu), a natural nutrient, could specifically facilitate the exocytosis of Glu-APD NPs, indicating an amino-acid-associated intracellular trafficking. Oral administration of insulin-loaded Glu-APD NPs could also achieve a good hypoglycemic effect with a relative bioavailability of 10.04%, which is 1.89-fold higher than that of P NPs and 5.20-fold higher than insulin solution. Safety evaluations further verified the biocompatibility of Glu-APD NPs and the related materials. The results confirmed the feasibility of introducing Glu-APD to NPs for improving the oral delivery of insulin. With the surface features of dendritic peptide, Glu-APD could facilitate oligopeptide/amino-acid-associated transport of the related NPs, which might be considered as an advantage under physiological conditions. This work might also be considered as a valid reference for the construction of highly efficient oral delivery systems.


 Please wait while we load your content...
Please wait while we load your content...