Surface modification of polymeric electrospun scaffolds via a potent and high-affinity integrin α4β1 ligand improved the adhesion, spreading and survival of human chorionic villus-derived mesenchymal stem cells: a new insight for fetal tissue engineering†
Abstract
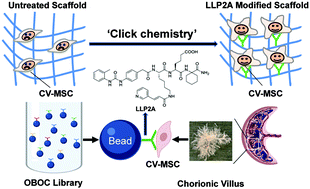
Cell–biomaterial interactions are primarily governed by cell adhesion, which arises from the binding of cellular integrins to the extracellular matrix (ECM). Integrins drive the assembly of focal contacts that serve as mechanotransducers and signaling nexuses for stem cells, for example integrin α4β1 plays pivotal roles in regulating mesenchymal stem cell (MSC) homing, adhesion, migration and differentiation. The strategy to control the integrin-mediated cell adhesion to bioinspired, ECM-mimicking materials is essential to regulate cell functions and tissue regeneration. Previously, using one-bead one-compound (OBOC) combinatorial technology, we discovered that LLP2A was a high-affinity peptidomimetic ligand (IC50 = 2 pM) against integrin α4β1. In this study, we identified that LLP2A had a strong binding to human early gestation chorionic villi-derived MSCs (CV-MSCs) via integrin α4β1. To improve CV-MSC seeding, expansion and delivery for regenerative applications, we constructed artificial scaffolds simulating the structure of the native ECM by immobilizing LLP2A onto the scaffold surface as cell adhesion sites. LLP2A modification significantly enhanced CV-MSC adhesion, spreading and viability on the polymeric scaffolds via regulating signaling pathways including phosphorylation of focal adhesion kinase (FAK), and AKT, NF-kB and Caspase 9. In addition, we also demonstrated that LLP2A had strong binding to MSCs of other sources, such as bone marrow-derived mesenchymal stem cells (BM-MSCs) and adipose tissue-derived mesenchymal stem cells (AT-MSCs). Therefore, LLP2A and its derivatives not only hold great promise for improving CV-MSC-mediated treatment of fetal diseases, but they can also be widely applied to functionalize various biological and medical materials, which are in need of MSC recruitment, enrichment and survival, for regenerative medicine applications.


 Please wait while we load your content...
Please wait while we load your content...