Preparation of photothermal-chemotherapy nanohybrids by complexation of gold nanorods with polyamidoamine dendrimers having poly(ethylene glycol) and hydrophobic chains†
Abstract
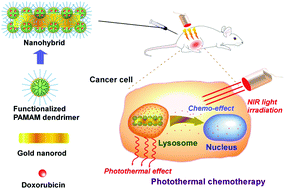
The combination of anticancer drugs and laser hyperthermia could lead to efficient cancer treatment with less-adverse effects. This study combined anticancer drug-loaded functional dendrimers and light-responsive gold nanorods to fabricate nanohybrids that can provide anticancer-drug delivery and subsequent heat generation under near-infrared laser irradiation. A condensation reaction was used to conjugate poly(ethylene glycol)-modified polyamidoamine dendrimers to carboxylated gold nanorod surfaces. Oleoyl groups were incorporated into dendrimers to improve the drug loading capacity. Doxorubicin loading capacity was improved by incorporation of oleoyl chains into dendrimers in the nanohybrid, indicating increased hydrophobic interaction between anticancer drugs and nanohybrids. The nanohybrids exhibited heat generation properties under near infrared laser irradiation. They released anticancer drugs over time. The combination of doxorubicin-loaded nanohybrids and laser irradiation showed markedly better cytotoxicity than that of the nanohybrids used with lasers and drug-loaded nanohybrids without the use of lasers. After intravenous or intratumoral injection of nanohybrids to tumor-bearing mice, a sharp temperature increase was observed at the tumor site under laser irradiation. Especially, intratumorally injected doxorubicin-loaded nanohybrids showed almost complete tumor growth suppression under laser irradiation. The results demonstrate that functional dendrimer–gold nanorod nanohybrids are promising as multi-functional nanomaterials to achieve synergistic effects of anticancer drugs and heat ablation to support effective cancer treatments.


 Please wait while we load your content...
Please wait while we load your content...