Shuttle/sink model composed of β-cyclodextrin and simvastatin-loaded discoidal reconstituted high-density lipoprotein for enhanced cholesterol efflux and drug uptake in macrophage/foam cells†
Abstract
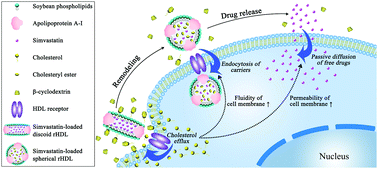
Targeting drug delivery to macrophage/foam cells is challenged owing to the poor cell permeability and fluidity resulting from the massive accumulation of intracellular cholesterol in atherosclerosis (AS). Discoidal reconstituted high-density lipoprotein (d-rHDL) has been well regarded as a potential drug delivery system for AS by virtue of its plaque-targeting and cholesterol removal abilities, while the latter is compromised by the high activation energy of cholesterol efflux. It is reported that a low concentration of β-cyclodextrin (β-CD) can function as a cholesterol shuttle to promote cholesterol efflux from cells to the extracellular acceptors (cholesterol sink, such as HDL particles), but it is still unknown whether the combination of β-CD with a drug-loaded d-rHDL can function as a shuttle/sink model to promote the remodeling and drug release of the d-rHDL carrier after accelerating the cholesterol efflux. Furthermore, it is interesting to investigate whether enhanced cholesterol efflux can improve the cellular drug uptake by restoring the permeability and fluidity of the cell membrane. Here, simvastatin-loaded d-rHDL (ST-d-rHDL) was combined with different concentrations of β-CD. Compared with ST-d-rHDL alone, the cholesterol removal ability of ST-d-rHDL combined with 0.5 mM of β-CD increased by 31-fold after incubation for 6 h and the cumulative drug release of ST-d-rHDL increased by two-fold during the initial 1 h in an acellular mimetic system. In macrophage/foam cells, 0.5 mM of β-CD showed superior promoting effects in the cholesterol removal ability and remodeling of ST-d-rHDL compared to 0.1 mM of β-CD. The high concentration of β-CD at 2 mM displayed a low efficiency for accelerating cholesterol efflux, which might function as a cholesterol sink rather than a cholesterol shuttle. Moreover, the permeability and fluidity of the cell membrane were improved by combining 0.5 mM of β-CD with ST-d-rHDL, which exhibited an enhanced cellular drug uptake and inhibiting effect on the intracellular lipid deposition and secretion of inflammatory cytokine. Collectively, combination of β-CD and ST-d-rHDL as a shuttle/sink model could enhance cholesterol efflux and drug uptake to suppress inflammation in macrophage/foam cells.


 Please wait while we load your content...
Please wait while we load your content...