Unravelling the origin of the photocarrier dynamics of fullerene-derivative passivation of SnO2 electron transporters in perovskite solar cells†
Abstract
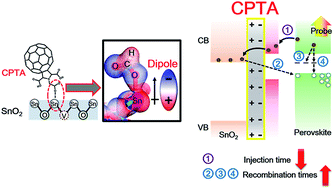
Fullerene-passivated SnO2 electron transport layers (ETLs) offer a route for a continuous boost in the power conversion efficiencies (PCEs) of perovskite solar cells (PSCs). However, a detailed understanding on the photocarrier dynamics in PSCs consisting of fullerene-passivated SnO2 ETLs is still lacking. Here, we use ultrafast pump-probe transient absorption spectroscopy to analyze the corresponding photocarrier dynamics across fullerene-modified SnO2 ETLs in PSCs. Both the C60 pyrrolidine tris-acid (CPTA)-modified SnO2 ETL and 6,6-phenyl C61-butyric acid methyl ester (PC61BM)-modified SnO2 ETL which have a favorable energy band edge alignment to perovskites, result in an enhanced electron injection rate after introducing these two fullerenes into SnO2-based PSCs compared to the SnO2 ETL without modification. Moreover, combining chemical measurements and density functional theory simulation results, it is confirmed that the interfacial chemical bond is tailored between the SnO2 surface and carboxylic acids of CPTA, thus establishing interface dipoles. Due to the existence of interface dipoles, the chemisorption of the CPTA fullerene functions as a recombination suppressor both at the CPTA-passivated SnO2/perovskite interface and inside perovskites. By contrast, the physisorption of the PC61BM fullerene on the SnO2 ETL exhibits relatively high recombination rates both at the SnO2/perovskite interface and inside perovskites. Therefore, the PSC based on the SnO2/CPTA ETL yields a PCE of more than 19%, which is superior to that of the reference PSC using PC61BM as a passivator (<18%). This study describes that interfacial carrier dynamics in PSCs consisting of fullerene-modified ETLs can be clearly resolved using ultrafast spectroscopy, which further provides guidance for future design for interfacial modification of ETLs.


 Please wait while we load your content...
Please wait while we load your content...