Monodisperse PdSn/SnOx core/shell nanoparticles with superior electrocatalytic ethanol oxidation performance†
Abstract
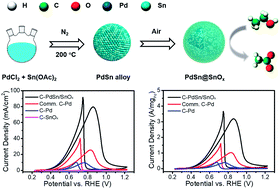
Direct ethanol fuel cells are one of the most promising electrochemical energy conversion devices for portable electronics and electric vehicles. Highly efficient and robust electrocatalysts for the ethanol oxidation reaction are therefore desired. In this paper, we report a facile approach for synthesizing monodisperse PdSn/SnOx core/shell nanoparticles as high-performance catalysts for electrochemical ethanol oxidation reaction (EOR). The mass current density of PdSn/SnOx reached 3.2 A mgPd−1 at 0.85 V vs. RHE in 1 M KOH solution, which is 3.2 times higher than that of the commercial Pd catalyst. More importantly, these PdSn/SnOx core/shell nanoparticles exhibit enhanced stability compared with the commercial Pd catalyst. Density functional theory calculations suggest that the strong d–p orbital coupling and sp-electron transfer in the PdSn alloy weaken the adsorption of carbonaceous species, e.g., the acetate-evolution intermediate *CH3CO and the poisoning species *CO, at Pd sites. Consequently, the destabilization of these species facilitates their coupling with *OH, leading to enhanced EOR activity and poison resistance. Meanwhile, we found that amorphous SnOx layers possess rich Sn–Sn ensembles with stronger adsorption of *OH, providing a shuttling path of hydroxide ions to surface sites.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...