Sonication-induced electrostatic assembly of an FeCO3@Ti3C2 nanocomposite for robust lithium storage†
Abstract
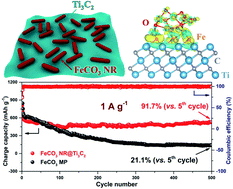
Transition metal carbonates as represented by FeCO3 have been widely evaluated as attractive anode alternatives for high-energy lithium ion batteries (LIBs), owing to their high practical lithium storage capacity approximately three times that of a conventional graphite anode. However, capacity-decay upon cycling and unsatisfactory rate performance have been the critical issues hindering their practical applications. We report here a structural reconstruction protocol, coupled with compositing with conductive MXene materials to address the above issues. Specifically, sonication-induced electrostatic assembly (SIEA) is exploited to convert a hybrid consisting of FeCO3 microparticles and multilayered Ti3C2 stacks into reshaped FeCO3 nanorods that are uniformly anchored at the surface of highly exfoliated Ti3C2 monolayers. Through the X-ray photoelectron spectroscopy (XPS) and zeta potential analysis, it was seen that the surface Fe2+ to Fe3+ oxidation plays a critical role in positively charging the particle surface of FeCO3 nanorods, resulting in desired composite construction with the negatively charged Ti3C2 substrate. Within such a composite material, charge transfer to active FeCO3 particles is effectively facilitated during lithiation/delithiation processes, which has been predicted by density functional theory (DFT) calculations and further verified by in situ electrochemical impedance spectroscopy (EIS) measurements. Concurrent implementation of nano-engineering and the MXene-support through the SIEA results in remarkably enhanced cycling stability and rate capability of the FeCO3 anode material. This work offers an effective material engineering strategy to boost the lithium storage performance for transition metal carbonate anode materials.


 Please wait while we load your content...
Please wait while we load your content...