Insights into the Li incorporation effect in Ni/Co-free P2-type Na0.6Mn0.8Cu0.2O2 for sodium-ion batteries†
Abstract
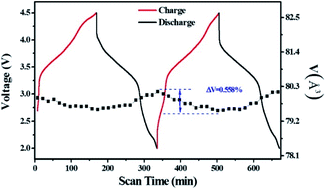
Copper-containing composites have been widely investigated among various sodium-layered cathode materials due to the low cost and abundance of the copper element. However, the electrochemical performance of these composites is usually deteriorated by the phase transitions corresponding to the stepwise charge/discharge curves, leading to an unsatisfactory capacity stability. Herein, we report that an improved electrochemical performance was achieved by substituting manganese with lithium in the Ni/Co-free P2-type Na0.6Mn0.8−xLixCu0.2O2−δ (x = 0, 0.05, 0.1, 0.15 and 0.2, δ > 0) composites with smooth charged/discharged curves. A capacity retention rate was increased from 54.3% to 82.8% over 200 cycles after adding 0.15 mole of lithium in per stoichiometric composites (Na0.6Mn0.65Li0.15Cu0.2O2−δ). Moreover, the rate performances were all enhanced in the lithium-incorporated composites compared to that of the pristine one. The interphase changes during and after cycling were studied by ex situ and in situ electrochemical impedance spectroscopy measurements, which prove that Li benefits the charge transfer and stabilizes the interphase of the Na0.6Mn0.65Li0.15Cu0.2O2−δ electrode. The in situ X-ray diffractometry results illustrate an alleviated crystallographic change in Na0.6Mn0.65Li0.15Cu0.2O2−δ compared to the Li-free electrode. These combined results provide explanations for the enhanced electrochemical performance after Li substitution in the Na0.6Mn0.8Cu0.2O2 composites from the aspect of the interfacial properties and structure variation. In addition, the online differential electrochemical mass spectrometry test demonstrates that the overall evolved CO2 gas in Na0.6Mn0.65Li0.15Cu0.2O2−δ cell is lower than that in the lithium-free cell during the initial cycle.


 Please wait while we load your content...
Please wait while we load your content...