Electron affinity regulation on ultrathin manganese oxide nanosheets toward ultra-stable pseudocapacitance†
Abstract
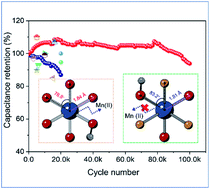
Although manganese oxide (MnO2) has long been considered as a promising electrode material for pseudocapacitors due to its high theoretical capacity and large potential window, its rapid capacity fading severely impedes its further large-scale applications. Herein, an electron affinity regulation strategy is developed to inhibit the dissolution of Mn2+ during the charging and discharging process. Remarkably, fluoride (F−) substituted MnO2 (MnOF) nanosheets exhibit an exceptionally high durability (no obvious degradation after 100 000 cycles even at a high scan rate of 200 mV s−1) along with an enhanced capacitance in an aqueous (Na2SO4) electrolyte, which is superior to that of all the reported MnOx electrodes and comparable to that of carbon-based electrodes. DFT calculations and X-ray fine structure characterization reveal that the non-equilibrium F substitution in MnO2 induces the enhanced energy barrier (ΔG) of the Mn(III) disproportionation reaction and greatly stabilizes the Mn–O bond, which are the key in boosting cycling life.


 Please wait while we load your content...
Please wait while we load your content...