A carbon–silica–zirconia ceramic membrane with CO2 flow-switching behaviour promising versatile high-temperature H2/CO2 separation†
Abstract
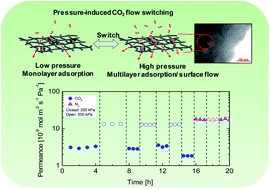
Many researchers regard silica, silica-based and zeolite membranes as the agents that will accomplish H2 separation. These membranes are expected to be productive in various mixture systems and under very high temperatures. This work reports the successful fabrication of a composite carbon–SiO2–ZrO2 ceramic membrane with a unique pressure-induced switching of CO2 flows that allows versatile H2/CO2 separation at elevated temperatures. TG-MS, DTG-TGA, FT-IR, CP-MAS-13C-NMR, and TEM provide corroborative evidence of the carbonization of starting material SiO2–ZrO2-acetylacetonate into C–SiO2–ZrO2. The resultant C–SiO2–ZrO2 displayed significant hysteresis in the CO2 adsorption isotherm at a temperature well above the critical temperature of CO2 (31 °C), which indicates structural conformation. Furthermore, single-gas permeation measurements showing upstream pressures of 200 and 500 kPa reveal different permeation values for CO2 at 300 °C. In separating a H2/CO2 mixture at 50 and 300 °C under upstream pressures of 200 and 500 kPa, respectively, the flow of H2 permeance reduces as the concentration of CO2 increases in the feed side at 50 °C (1.14 × 10−8 down to 3.9 × 10−9 mol m−2 s−1 Pa−1 at 200 kPa). The pressure-induced surface flow of CO2 at 300 °C and 500 kPa, however, reduces the hindrance to H2 flow and results in H2/CO2 selectivity of ∼20–30 for all CO2 concentrations, which is on a par with molecular sieving membranes. This novel C–SiO2–ZrO2 material shows promise for many interesting applications.


 Please wait while we load your content...
Please wait while we load your content...