Revealing the effect of polyethylenimine on zinc metal anodes in alkaline electrolyte solution for zinc–air batteries: mechanism studies of dendrite suppression and corrosion inhibition†
Abstract
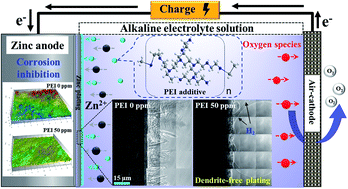
Zinc batteries have great potential as future energy storage devices due to their low cost and high energy density. Electrolyte additives have been developed for perfecting the performance of zinc batteries recently. In this work, the effects of polyethylenimine (PEI) additive on dendrite suppression and corrosion inhibition of the zinc anode in alkaline electrolyte solution are studied. The morphological evolution and interfacial reaction on the zinc anode with the addition of PEI are investigated via in operando visualization by both transmission X-ray microscopy (TXM) and optical microscopy (OM) for the first time. In zinc plating, from the in operando TXM images, the deposited zinc nuclei are changed from a spiky dendritic structure to a dense film as PEI concentration increases, and the morphology of nuclei dominates the structural evolution of zinc deposits later on. Meanwhile, an enhancement of interfacial hydrogen evolution on the zinc anode in the presence of PEI is observed by in operando OM. The addition of PEI also efficiently suppresses the self-corrosion damage of the zinc anode. The inhibition efficiency of zinc anode corrosion reaches up to 52.2% in the electrolyte with 50 ppm PEI, and the surface roughness of the corroded zinc anode is significantly reduced. In addition, a zinc–air full cell with PEI additive exhibits good cycling stability at current densities of 5 mA cm−1 and 10 mA cm−1, which can be attributed to the mitigation of electrode deterioration during cycling. The proposed mechanisms reveal the beneficial effects of PEI on planar nucleus growth and zinc corrosion protection, and the potential for practical application in zinc–air batteries.


 Please wait while we load your content...
Please wait while we load your content...