In operando synchrotron X-ray studies of a novel spinel (Ni0.2Co0.2Mn0.2Fe0.2Ti0.2)3O4 high-entropy oxide for energy storage applications†
Abstract
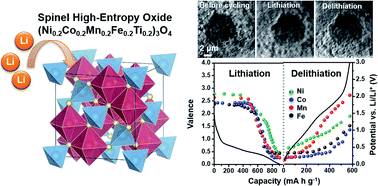
High-entropy oxides (HEOs) consisting of multiple cations have garnered considerable attention in recent years because of their unique structures and functional properties, which have enabled various applications. An entropy-stabilized rock-salt-based HEO, namely, (Mg0.2Co0.2Ni0.2Cu0.2Zn0.2)O, has recently been proposed as an anode material for lithium-ion batteries, and has exhibited promising features for energy storage applications with high capacity and stability. In this study, we utilized a simple solid-state sintering method to synthesize a single-phase spinel-structured HEO, (Ni0.2Co0.2Mn0.2Fe0.2Ti0.2)3O4 (NCMFT), for the first time. As an anode material for lithium-ion batteries, NCMFT has a high capacity (∼560 mA h g−1) at a current density of 100 mA g−1 and exhibits an excellent capacity retention of 100% after 100 cycles. Through in operando synchrotron X-ray absorption near edge structure and ex situ X-ray photoelectron spectroscopy analyses, we can understand the redox reactions that occur in the NCMFT anode during lithiation and delithiation. In operando synchrotron X-ray diffraction and ex situ transmission electron microscopy were used to analyze structural changes during the conversion reactions. In addition, the volume change behavior of the NCMFT anode was observed through in operando synchrotron transmission X-ray microscopy. Through the aforementioned analyses, the energy storage mechanism of NCMFT anodes was systematically investigated. This work provides various essential methods for understanding the mechanism of HEOs in energy storage applications.


 Please wait while we load your content...
Please wait while we load your content...