An innovative approach to design SOFC air electrode materials: high entropy La1−xSrx(Co,Cr,Fe,Mn,Ni)O3−δ (x = 0, 0.1, 0.2, 0.3) perovskites synthesized by the sol–gel method†
Abstract
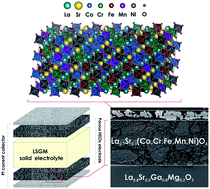
Among the for the first time reported Cr-containing high entropy La1−xSrx(Co,Cr,Fe,Mn,Ni)O3−δ (x = 0, 0.1, 0.2, 0.3, 0.4 and 0.5) perovskite-type oxides, the selected Sr-doped La0.7Sr0.3(Co,Cr,Fe,Mn,Ni)O3−δ material is documented to possess attractive properties as a candidate air electrode material for Solid Oxide Fuel Cells (SOFCs). Nanosized powders of the considered oxides are obtained using a modified Pechini sol–gel method. In the formed solid solution with a simple perovskite structure the strontium solubility limit is found to be at least x = 0.3. Room temperature (RT) structural data indicate the presence of rhombohedral structural distortion (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c symmetry) in the materials. High-temperature structural studies for the selected La0.7Sr0.3(Co,Cr,Fe,Mn,Ni)O3−δ indicate the occurrence of a phase transition to an aristotype Pm
c symmetry) in the materials. High-temperature structural studies for the selected La0.7Sr0.3(Co,Cr,Fe,Mn,Ni)O3−δ indicate the occurrence of a phase transition to an aristotype Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m structure at ca. 800 °C. The linear thermal expansion coefficient in the RT-1000 °C range is found to be moderate, 16.0(3) × 10−6 K−1. The results of impedance spectroscopy measurements support the semiconducting-type behavior of the electrical conductivity for all single-phase materials, in a temperature range of RT-1000 °C. The maximum recorded conductivity for the La0.7Sr0.3(Co,Cr,Fe,Mn,Ni)O3−δ composition exceeds 16 S cm−1 in the 900–1000 °C range, being suitable for application. Furthermore, chemical stability toward the La0.8Sr0.2Ga0.8Mg0.2O3−δ (LSGM) electrolyte is proven. Considering the presence of chromium, typically deleterious to the performance, the measured value of the total cathodic polarization resistance for the La0.7Sr0.3(Co,Cr,Fe,Mn,Ni)O3−δ-based electrode, being 0.126 Ω cm−2 at 900 °C, seems to be very attractive. The results obtained for a button-type fuel cell indicate power densities at a level of 550 mW cm−2 at 900 °C. Therefore, it can be considered that the high entropy-based approach enables to propose alternative SOFC air electrode materials, with otherwise inaccessible chemical compositions.
m structure at ca. 800 °C. The linear thermal expansion coefficient in the RT-1000 °C range is found to be moderate, 16.0(3) × 10−6 K−1. The results of impedance spectroscopy measurements support the semiconducting-type behavior of the electrical conductivity for all single-phase materials, in a temperature range of RT-1000 °C. The maximum recorded conductivity for the La0.7Sr0.3(Co,Cr,Fe,Mn,Ni)O3−δ composition exceeds 16 S cm−1 in the 900–1000 °C range, being suitable for application. Furthermore, chemical stability toward the La0.8Sr0.2Ga0.8Mg0.2O3−δ (LSGM) electrolyte is proven. Considering the presence of chromium, typically deleterious to the performance, the measured value of the total cathodic polarization resistance for the La0.7Sr0.3(Co,Cr,Fe,Mn,Ni)O3−δ-based electrode, being 0.126 Ω cm−2 at 900 °C, seems to be very attractive. The results obtained for a button-type fuel cell indicate power densities at a level of 550 mW cm−2 at 900 °C. Therefore, it can be considered that the high entropy-based approach enables to propose alternative SOFC air electrode materials, with otherwise inaccessible chemical compositions.
- This article is part of the themed collection: Editor’s Choice: Solid-state ion conductors


 Please wait while we load your content...
Please wait while we load your content...