A facile reflux method for in situ fabrication of a non-cytotoxic Bi2S3/β-Bi2O3/ZnIn2S4 ternary photocatalyst: a novel dual Z-scheme system with enhanced multifunctional photocatalytic activity†
Abstract
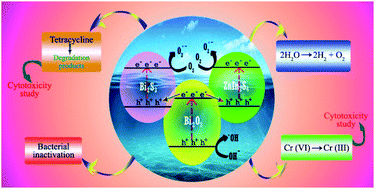
In this study, we have developed a novel non-cytotoxic Bi2S3/β-Bi2O3/ZnIn2S4 ternary nanocomposite using a mild in situ reflux method for multimodal applications in the fields of waste water remediation, H2 energy production and microbial disinfection. Initially, the metastable β-Bi2O3 phase with nanoplate morphology was synthesized by thermal decomposition of Bi2O2CO3. The co-assembly of Bi2S3 and ZnIn2S4 with β-Bi2O3 is achieved by a tailor made in situ reflux route using thioacetamide as the sulfur source. Comprehensive characterization of the ternary composite revealed close microscopic contact between the semiconductors, fast electron channelization, enhanced charge carrier separation and a prolonged life time (11.25 ns) of the excited state. The ternary composite exhibits excellent visible light assisted photocatalytic activity for aqueous phase tetracycline (TCN) degradation and Cr(VI) reduction achieving 96.3% (Kapp = 0.0868 min−1) and 93.7% (Kapp = 0.2236 min−1) removal in a short reaction span of 40 min and 12 min, respectively. The ternary composite also afforded a H2 generation rate of 2243 μmol h−1 g−1 with an apparent conversion efficiency of 14.3%. A bacterial inactivation study conducted using Enterobacter cloacae suggested good visible light assisted bacterial degradation activity of the coupled semiconductor system. The MTT assay study confirms the non-cytotoxic nature of the photocatalyst as well as the treated TCN and Cr(VI) solutions. A synergistic dual Z-scheme electron migration mechanism is deduced from radical trapping experiments and the ESR study which accounts for the excellent photocatalytic activity of the ternary composite material.


 Please wait while we load your content...
Please wait while we load your content...