Novel approaches for highly selective, room-temperature gas sensors based on atomically dispersed non-precious metals†
Abstract
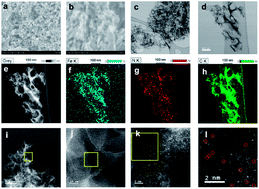
Atomically dispersed (AD) materials have incredible catalytic ability and offer atom economy with 100% metal utilization during catalytic reactions. Herein, we report the first attempt to synthesize AD FeNC materials for use as highly selective, room-temperature gas sensors. Aberration-corrected high-angle annular dark-field scanning transmission electron microscopy (AC-HAADF-STEM), extended X-ray absorption fine structure (EXAFS) spectroscopy, and Mössbauer spectroscopy characterization methods confirm the existence of atomically dispersed Fe with an FeN4 coordination. We demonstrate a room temperature, sensitive, and selective NO2 gas sensor technology using this platform, offering significant advantages over existing technology. Density functional theory (DFT) calculations verify that electrons are transferred to NO2 from FeN4 during NO2 adsorption. Both DFT calculations and experiments also reveal that the barrier for NO2 decomposition by AD FeNC is 0.73 eV, which is significantly lower than previously reported barriers (2.60–3.54 eV) in other materials. Such unique catalytic properties combined with a high surface affinity for NO2 molecules enable AD FeNC gas sensors to have excellent selective NO2 detection at room temperature. The method proposed here can inspire the use of AD materials to detect other gases and catalyze similar novel developments in the gas sensor industry.


 Please wait while we load your content...
Please wait while we load your content...