Anode potential controlled charging prevents lithium plating
Abstract
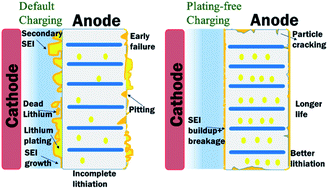
We report a novel anode potential controlled charging strategy for lithium-ion cells which eliminates lithium plating under most aggressive conditions, such as at low temperatures. This is applicable for lithium-ion cells with a graphite anode irrespective of the form factor, capacity or cathode chemistry. This new charging strategy leads to a seven-fold increase in cycle life and a concomitant improvement in the electrochemical performance. Conventional charging shows copious lithium plating swiftly followed by cell failure due to accelerated increase in the anode resistance. The anode potential controlled charging strategy, based on a three-electrode cell construction, exhibits minimal increase in the anode resistance and shows no signs of lithium plating in operational extremes. Optical micrographs and high-resolution scanning electron images confirm that the graphite anode in the conventionally charged Li-ion cell undergoes significant loss in porosity resulting in massive underlithiation and dramatic capacity fading. The degradation rate in the anode is decelerated in anode potential controlled charging by ensuring that plating potential is not reached and improving ion transport in the anode even at low temperatures due to the absence of decomposition products that would have formed during plating.


 Please wait while we load your content...
Please wait while we load your content...