Highly conductive fluorine-based anion exchange membranes with robust alkaline durability†
Abstract
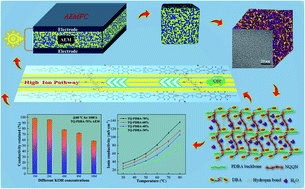
Anion exchange membranes (AEMs) with robust alkaline stability and high ionic conductivity are imminently required for the promising electrochemical energy conversion devices – fuel cells. Herein, a series of novel crosslinked AEMs with hydrophobic fluorine-based polymer backbones bearing special functional sites and hydrophilic long flexible multi-cation side chains are prepared. Morphology observation and ion transport analysis confirm the existence of distinct microphase separation and efficient ion-conducting channels within the membranes resulting from the inherent chemical structure. A highest ionic conductivity of 136.27 mS cm−1 can be achieved by TQ-PDBA-70% (IEC = 2.16 meq. g−1) at 80 °C. Meanwhile, the prepared TQ-PDBA-X AEMs exhibit a desirable swelling ratio (<10%) and excellent mechanical properties (tensile stress > 22.8 MPa). It is worth noting that the retained ionic conductivity of the TQ-PDBA-70% AEM is 98.14%, 95.50%, 77.90%, 72.02% and 58.15% after being immersed in 1, 2, 4, 8 and 10 M KOH at 80 °C for 1000 h, respectively. Chemical structure change of the TQ-PDBA-70% AEM before and after the alkaline stability test is negligible, as revealed by FT-IR. Moreover, TQ-PDBA-70% has high ionic exchange capacity (IEC) retention and maintains good mechanical properties. A single cell assembled with TQ-PDBA-70% has a maximum power density of 158.8 mW cm−2 under a current density of 360 mA cm−2. These results suggest that this type of structure opens a new strategy for developing high performance AEMs.


 Please wait while we load your content...
Please wait while we load your content...