Fabrication of CuOx nanowires@NiMnOx nanosheets core@shell-type electrocatalysts: crucial roles of defect modification and valence states for overall water electrolysis†
Abstract
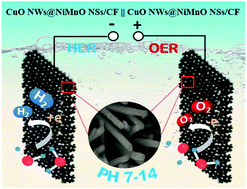
Combining oxides with bimetallic elements may be an efficient route to modify the electronic distribution for improving the intrinsic activity of electrocatalysts, and further engineering the defect structure and adjusting the valence state could provide more adsorption sites as well as active sites for the decomposition of reactants and/or intermediates. Herein, NiMnOx nanosheets with rich defects were electrochemically deposited on the surface of Cu(OH)2 nanowires followed by a thermal treatment process to form a CuOx nanowires@NiMnOx nanosheets (CuOx NWs@NiMnOx NSs) electrocatalyst with a core–shell configuration. Structural characterization indicated that NiMnOx nanosheets had numerous vacancies with scattered defects and dislocations with partial cracking of the inert basal planes, resulting in extra active edge sites and a coordination-unsaturated spinel crystal. As such, the valence states of Ni and Mn elements became unstable in the defect modified NiMnOx materials under the applied potential. Compared with the initial NiMnOx, more Ni3+ and Mn4+ were formed during the oxygen evolution reaction (OER) whereas they were converted to Ni2+ and Mn3+ during the hydrogen evolution reaction (HER). These controllable valence state changes of metal elements endowed the NiMnOx with high ability for catalyzing overall water electrolysis. As a result, the CuOx NWs@NiMnOx NSs bifunctional electrocatalysts exhibited high performances in both HER and OER in an alkaline electrolyte. Meanwhile, by using the same electrocatalysts in a neutral electrolyte, overpotentials at 10 mA cm−2 were also as low as 80.7 and 390 mV with Tafel slopes of 77.6 and 101.6 mV dec−1 for the HER and OER, respectively. A two-electrode overall water electrolysis system using this bifunctional electrocatalyst exhibited low cell voltages of 1.62 and 1.75 V in alkaline and neutral electrolytes respectively at the standard current density of 10 mA cm−2 with long-term stability, indicating that this CuOx NWs@NiMnOx NSs core@shell-type material should be an effective bifunctional electrocatalyst in both alkaline and neutral pH media.


 Please wait while we load your content...
Please wait while we load your content...