An ultrathin Al2O3 bridging layer between CdS and ZnO boosts photocatalytic hydrogen production†
Abstract
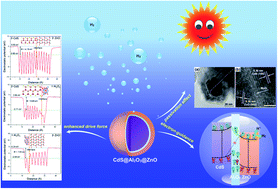
The rapid separation and transfer of photogenerated charge carriers are the essential criteria for high photocatalytic efficiency. Heterostructures (HSs) composed of several semiconductors can take advantage of energy level difference to promote the separation and transfer of photogenerated electrons and holes, where the interface between semiconductors is the most critical position which directly determines the transfer mechanism of photogenerated charge carriers, and ultimately determines the photocatalytic efficiency. Herein, an ultrathin Al2O3 bridging layer is intentionally introduced into the interface between CdS and ZnO by using an atomic layer deposition (ALD) method, and the resultant CdS@Al2O3@ZnO catalyst exhibits a significantly enhanced H2 evolution rate (1190 μmol h−1 m−2) and a high apparent quantum efficiency (AQE, 31.14% at a wavelength of 420 nm). It is revealed that the ultrathin Al2O3 bridging layer converts the original Z-scheme mechanism of photogenerated charge carriers in the CdS@ZnO catalyst into a modified type II mechanism in the CdS@Al2O3@ZnO catalyst, where the photogenerated electrons are tunnelled from the CdS core to the ZnO shell and then are depleted by H2O effectively in time to produce H2. Meanwhile, the photogenerated holes are consumed efficiently by the fixed negative charges of Al2O3. Detailed characterization reveals that the lattice mismatch between wurtzite ZnO and wurtzite CdS is effectively passivated by the Al2O3 interlayer, facilitating the uniform deposition of the ZnO layer, which promotes the close contact between interfaces, suppressing the recombination of charge carriers. This work provides a new strategy for controlling the dominant transfer mechanisms of photogenerated electrons and holes in HSs through interface regulation.


 Please wait while we load your content...
Please wait while we load your content...