Do polymer ligands block the catalysis of metal nanoparticles? Unexpected importance of binding motifs in improving catalytic activity†
Abstract
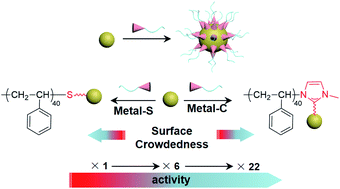
Metal nanoparticles (NPs) tethered by synthetic polymers are of broad interest for self-assembly, nanomedicine and catalysis. The binding motifs in polymer ligands usually as the end functional groups of polymers are mostly limited to thiolates. Since the binding motif only represents a tiny fraction of many repeating units in polymers, its importance is often ignored. We herein report the uniqueness of polymeric N-heterocyclic carbene (NHC) ligands in providing oxidative stability and promoting the catalytic activity of noble metal NPs. Two “grafting to” methods were developed for polymer NHCs for pre-synthesized metal NPs in various solvents and with different sizes. Remarkably, imidazolium-terminated polystyrene can modify gold NPs (AuNPs) within 2 min while reaching a similar grafting density to polystyrene-thiol (SH) requiring 6 h modification. We demonstrate that polymer NHCs are extremely stable at high temperature in air. Interestingly, the binding motifs of polymer ligands dominate the catalytic activity of metal NPs. Polymer NHC modified metal NPs showed improved activity regardless of the surface crowdedness. In the case of AuNPs, AuNPs modified with polystyrene NHCs are approximately 5.2 times more active than citrate-capped ones and 22 times more active than those modified with polystyrene thiolates. In view of ligand-controlled catalytic properties of metal NPs, our results illustrate the importance of binding motifs that has been overlooked in the past.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...