Evaluation of sputtered nickel oxide, cobalt oxide and nickel–cobalt oxide on n-type silicon photoanodes for solar-driven O2(g) evolution from water†
Abstract
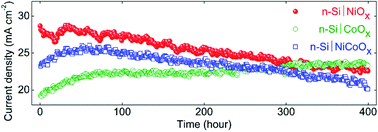
Thin films of nickel oxide (NiOx), cobalt oxide (CoOx) and nickel–cobalt oxide (NiCoOx) were sputtered onto n-Si(111) surfaces to produce a series of integrated, protected Si photoanodes that did not require deposition of a separate heterogeneous electrocatalyst for water oxidation. The p-type transparent conductive oxides (p-TCOs) acted as multi-functional transparent, antireflective, electrically conductive, chemically stable coatings that also were active electrocatalysts for the oxidation of water to O2(g). Relative to the formal potential for water oxidation to O2, Eo′(O2/H2O), under simulated Air Mass (AM)1.5 illumination the p-TCO-coated n-Si(111) photoanodes produced mutually similar open-circuit potentials of −270 ± 20 mV, but different photocurrent densities at Eo′(O2/H2O), of 28 ± 0.3 mA cm−2 for NiOx, 18 ± 0.3 mA cm−2 for CoOx and 24 ± 0.5 mA cm−2 for NiCoOx. The p-TCOs all provided protection from oxide growth for extended time periods, and produced stable photocurrent densities from n-Si in 1.0 M KOH(aq) (ACS grade) under potential control at Eo′(O2/H2O) for >400 h of continuous operation under 100 mW cm−2 of simulated AM1.5 illumination.


 Please wait while we load your content...
Please wait while we load your content...
