Enhancing CO2 reduction by suppressing hydrogen evolution with polytetrafluoroethylene protected copper nanoneedles†
Abstract
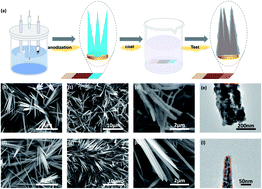
With the fast development of society and industry, atmospheric levels of carbon dioxide (CO2) have increased seriously, becoming a threat to the world’s climate. Electrochemical transformation of CO2 into fuels and chemicals using copper (Cu)-based materials has attracted enormous attention. However, the competitive hydrogen evolution reaction (HER) heavily influences their efficiency. Thus, it is urgent to promote the CO2 reduction reaction (CO2RR) and suppress the competitive HER. In this work, enhanced CO2RR with suppressed HER was achieved on polytetrafluoroethylene (PTFE) coated Cu nanoneedles (CuNNs). The concentration of surface adsorbed CO2 could be enhanced via the field-induced reagent concentration (FIRC) effect through the CuNN structures. The hydrophobic PTFE can prevent the supply of protons to CuNNs and thus suppress the HER. Scanning electron microscopy (SEM), transmission electron microscopy (TEM), and X-ray absorption spectroscopy (XAS) revealed that the PTFE coated CuNNs maintained the nanoneedle structures and metallic Cu state during the catalytic reaction process. As a result, highly suppressed HER coupled with high C2 selectivity can be achieved on these PTFE coated CuNNs with a Faraday efficiency (FE) of 47% toward C2 products and an ultralow FE of 5.9% toward H2 at −1.49 V vs. RHE (without IR correction). This work provides an effective strategy to promote the CO2RR and suppress the competitive HER.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...