A robust and highly active hydrogen evolution catalyst based on Ru nanocrystals supported on vertically oriented Cu nanoplates†
Abstract
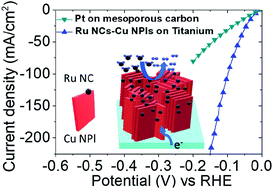
Pt nanoparticles deposited on mesoporous carbon (Pt/C) represent a benchmark catalyst for the hydrogen evolution reaction (HER) in both acid and alkaline conditions. However, it shows an order of magnitude lower activity under alkaline media compared to acid media. Moreover, the activity of this catalyst decreases rapidly under operational conditions due to both migration and aggregation of the metal particles, leading to a rapid decline in the number of active sites. We show here a Cu and Ru-based catalyst fabricated by slow electrodeposition of Ru onto a nanostructured Cu layer grown, in turn, on the top of a Ti substrate. Our catalyst has a high and stable HER activity, outperforming the benchmark Pt/C catalyst under alkaline conditions. The high activity and prolonged stability are attributed to the peculiar structure of our electrode, in which Cu nanoplates are directly attached perpendicular to the Ti substrate, providing a high surface area support onto which Ru nanocrystals are bound. This makes the Cu–Ru/Ti electrode one of the most active (−10 mA cm2 of HER current at only −23 mV overpotential) and stable electrocatalysts (no activity degradation for 100 h at −0.2 V) reported to date for the alkaline hydrogen evolution reaction.


 Please wait while we load your content...
Please wait while we load your content...