Surface-functionalized palladium catalysts for electrochemical CO2 reduction†
Abstract
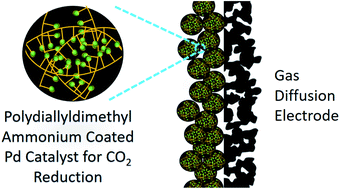
Rational design and synthesis of efficient catalysts for electrochemical CO2 reduction is a critical step towards practical CO2 electrolyzer systems. In this work, we report a strategy to tune the catalytic property of a metallic Pd catalyst by coating its surface with a polydiallyldimethyl ammonium (PDDA) polymer layer. The resulting PDDA-functionalized Pd/C catalysts exhibit an enhanced CO faradaic efficiency of ∼93% together with a current density of 300 mA cm−2 at −0.65 V versus reversible hydrogen electrode in comparison to non-functionalized and commercial Pd/C catalysts. X-ray photoelectron spectroscopy analysis reveals that the improvement can be attributed to the electron transfer from the quaternary ammonium groups of PDDA to Pd nanoparticles, weakening the CO binding energy on Pd. The weak CO adsorption on Pd was further confirmed by the CO temperature programmed desorption measurement and operando attenuated total reflection-Fourier-transform infrared analysis. Therefore, the incorporation of electron-donating groups could be an effective strategy to decrease the CO binding energy of a metallic catalyst for a high CO selectivity in CO2 electroreduction.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...