Tuning on and off chemical- and photo-activity of exfoliated MoSe2 nanosheets through morphologically selective “soft” covalent functionalization with porphyrins†
Abstract
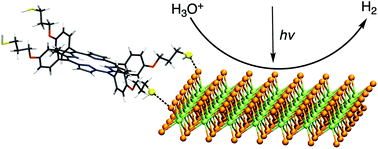
The covalent functionalization of 2D transition metal dichalcogenides (TMDCs) with organic molecules exploiting the thiol conjugation approach is still a very controversial topic. We have synthesized thiolated tetraphenyl porphyrins, with and without hydroxyl groups in the phenyl substituents, which have been covalently attached to chemically exfoliated MoSe2 (ce-MoSe2) nanosheets. XPS and FTIR have revealed the formation of sulfo-selenide bridges, specifically on the edges of the ce-MoSe2, as confirmed by first principles calculations. The electrocatalytic activity of TMDCs in the hydrogen evolution reaction (HER) has been investigated after conjugation with the organic molecules. The HER activity is suppressed or enhanced according to the presence of mildly acidic hydroxyl groups in the attached molecules, since they provide a local proton relay boosting the production of H2, especially under mildly acidic conditions (pH = 4.3). Moreover, the well-known light-harvesting properties of porphyrins have been exploited to improve significantly the light-assisted HER activity. Due to the formation of a type II heterojunction or Schottky contact between the molecules and the 2H and 1T MoSe2 nanosheets respectively, the hybrid materials show an improvement of the HER onset potential under illumination compared to the pristine material, without activity loss for more than 16 h.


 Please wait while we load your content...
Please wait while we load your content...