Mixed conductive composites for ‘Low-Temperature’ thermo-chemical CO2 splitting and syngas generation†
Abstract
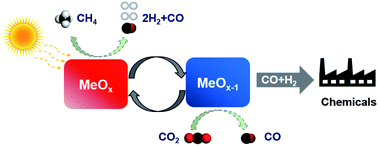
An effective strategy to design platinum group metal (PGM) free redox catalysts for “low temperature” CO2 splitting followed with methane partial oxidation was proposed and validated. Composites of mixed ionic-electronic conductive (MIEC) oxides were found to be highly effective at relatively low temperatures (600–750 °C). Specifically, perovskite structured LaNi0.35Fe0.65O3 and rock salt structured Ce0.85Gd0.1Cu0.05O2−δ, as two compatible yet structurally distinct MIEC oxides, were integrated into composite redox catalyst particles. Resulting from the synergistic effect of the two MIEC phases, 90% CO2 to CO conversion was demonstrated at 750 °C. Up to 90% methane conversion with 96% CO selectivity was also achieved in the methane POx step. The redox catalysts were characterized in detail to illustrate the underlying mechanisms for the synergistic effects. Electrical conductivity relaxation (ECR) measurements indicated significantly lowered activation energy for lattice oxygen (O2−) migration (0.43 eV). The enhanced oxygen migration in turn led to reversible exsolution of active transition metal nanoparticles (Ni–Fe alloy) from the mixed oxide, serving as active sites for methane activation while further enhancing lattice oxygen exchange, as confirmed by in situ X-ray diffraction and transmission electron microscopy. As a result, the composite redox catalysts demonstrate superior redox activity, coke resistance, and long term redox stability, making them potentially suitable for CO2 utilization and methane partial oxidation under a hybrid redox process scheme.


 Please wait while we load your content...
Please wait while we load your content...
