Ultrathin RuRh@(RuRh)O2 core@shell nanosheets as stable oxygen evolution electrocatalysts†
Abstract
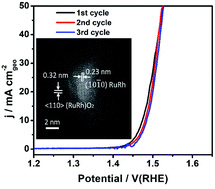
The oxygen evolution reaction (OER) is of prime importance in the development of highly efficient electrochemical energy conversion and storage technologies such as water splitting and zinc air batteries. Ru-based nanocatalysts are still the best catalytic system for the OER; however, one of the greatest challenges is their relatively poor stability for the OER. Here, we report a novel architecture of ultrathin RuRh@(RuRh)O2 core/shell nanosheets with a core of ultrathin metallic RuRh nanosheets and a shell of RuO2/RhO2 oxides as superb electrocatalysts toward the OER. The RuRh@(RuRh)O2 core/shell nanosheets can achieve a high electrocatalytic activity toward the OER, with a low 245 mV overpotential at 10 mA cm−2 and a Tafel slope of 51.2 mV dec−1, better than most state-of-the-art Ru-based or Ir-based electrocatalysts for the OER. Moreover, the RuO2/RhO2 oxides shell protect the normally labile RuRh NSs core against dissolution during the OER process, revealed by detailed rotating ring disk electrode (RRDE) measurements for simultaneously recording the dissolution of Ru on the ring electrode and the ex situ measurement of Ru dissolution using the galvanostatic method. Therefore, the RuRh@(RuRh)O2 core/shell nanosheets exhibit much better OER stability in acid media compared with RuRh nanosheets and commercial RuO2. Theoretical calculations reveal that once the formation of the surface oxidation layers is achieved, the O-sites evidentially crossover the Fermi level which could guarantee the high electroactivity towards adsorbates while the RuRh core serves as an electron reservoir with high electrical conductivity. The synergistic effect between the core and shell structure leads to the superior OER performance.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...