Hybrid capacitor with anthraquinone-grafted carbon as a battery-type electrode operating in a low pH aqueous salt solution
Abstract
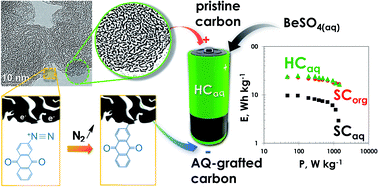
An all carbon battery-like material (SC2A@AQ) was developed by chemical grafting of anthraquinone (AQ) on high specific surface area carbon black (SC2A). A high degree of functionalization was gained owing to the enhanced number of active sites prone to readily react with diazonium terminated AQ cations. An excellent reversibility of the two-proton–two-electron transformation of the AQ moieties to 9,10-dihydroxyanthracene ones was achieved using a mild acidic medium (pH = 2.1) based on BeSO4. Accordingly, an aqueous hybrid capacitor (HCaq) was designed by combining SC2A@AQ, as a faradaic negative electrode, with its non-modified SC2A counterpart as a positive electrical-double layer one. During constant current discharge, from 0 to 1.5 V, the HCaq displays a quasi-linear profile and twice higher capacitance compared to the symmetric SC2A/SC2A capacitor (SCaq), viz. 51 vs. 24 F g−1 (per total active mass of the two electrodes), as well as considerably higher specific energy, viz. 15.8 vs. 6.2 W h kg−1 at a power of 1.0 kW kg−1. The capacitance retention of the HCaq reaches 87% after 10 000 galvanostatic cycles up to 1.6 V. Owing to the AQ grafting, the negative SC2A@AQ electrode of the HCaq demonstrates a very low potential decay (so-called self-discharge) of 0.11 V after a 4 hour voltage hold at 1.5 V, as opposed to 0.92 V for the SC2A negative electrode in the SCaq. Overall, the HCaq delivers energy and power performance comparable with a symmetric SC2A/SC2A capacitor operating up to 2.7 V in the usual organic electrolyte (SCorg), whilst it succeeds by being an economically viable energy storage device.


 Please wait while we load your content...
Please wait while we load your content...