Identifying the electrocatalytic active sites of a Ru-based catalyst with high Faraday efficiency in CO2-saturated media for an aqueous Zn–CO2 system†
Abstract
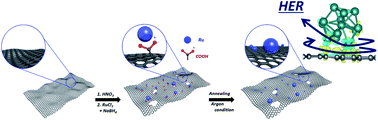
Developing a cost-effective, scalable, and efficient electrocatalyst for the hydrogen evolution reaction (HER) is the heart of producing pure hydrogen in practical applications. In this work, a unique approach to allow ruthenium (Ru) nanoparticles on carboxyl-functionalized porous sphere carbon (CF-Ru@PSC) for an aqueous Zn–CO2 system is reported. The CF-Ru@PSC catalyst was produced by inducing additional junction sites between metallic Ru and the carbon substrate for enhanced HER properties under a CO2 saturated condition. The strongly bonded Ru–C caused electron transfer from the bottom side of the metallic Ru to the PSC substrate, leading to efficient hydrogen adsorption energy of the surface. The DFT calculations and XAFS investigation suggested that the reconstructed electronic configuration from the Ru–C linkage acted as an efficient active site for HER. The CF-Ru@PSC electrocatalyst presented a high turnover frequency of 2.70H2 s−1 at an overpotential of 0.20 V in the CO2-saturated condition and high Faraday efficiency (98.2%) over 1000 min in the aqueous Zn–CO2 system.
- This article is part of the themed collection: Journal of Materials Chemistry A Lunar New Year collection 2021


 Please wait while we load your content...
Please wait while we load your content...