Highly selective aerobic oxidation of methane to methanol over gold decorated zinc oxide via photocatalysis†
Abstract
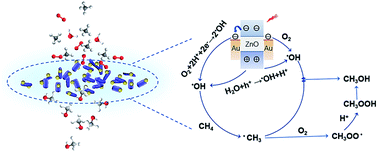
Photodriven CH4 conversion has been regarded as a promising green strategy for fabrication of value-added commodity chemicals, in particular methanol. However, due to the incomplete transformation of intermediates or the overoxidation of products, a good selectivity of methanol is hard to achieve. Here, we present a highly selective transformation of methane to methanol using gold modified zinc oxide as a photocatalyst under full light spectrum irradiation at atmospheric temperature. The selectivity of methanol can reach 99.1% with a productivity of 1371 μmol g−1. Fine tuning the loading amount of gold nanoparticles (0.75 wt%) and inputting an appropriate light energy are the pivotal factors for selectivity improvement. Besides, in contrast to the reported photocatalytic aerobic CH4 oxidation on gold modified zinc oxide, we find that both oxygen and water, rather than only molecular oxygen, provide the O-source for methanol formation. This result is verified through 18O-isotope tests (18O2 and H218O), leading to a disparate mechanism.


 Please wait while we load your content...
Please wait while we load your content...