Ultrathin AgPt alloy nanorods as low-cost oxygen reduction reaction electrocatalysts in proton exchange membrane fuel cells†
Abstract
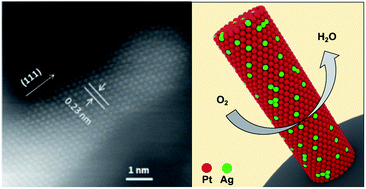
The excellent catalytic activities and stability of one-dimensional (1D) AgPt alloy nanostructures have been well reported toward the oxygen reduction reaction (ORR). However, their real application in proton exchange membrane fuel cells (PEMFCs) is still highly challenging due to the great difficulties in their preparation and fabrication into practical electrodes. By a facile self-growth assisted reduction method, ultrathin single-crystal AgPt alloy nanorods (NRs) with a diameter of 3–4 nm are uniformly grown on a carbon support through a finely controlled ion reduction process tuning the nucleation and growth of Pt and Ag. Enhanced power performance is successfully demonstrated in the single-cell test. With 50 at% Ag, the AgPt NR/C electrode shows 1.16-fold power density and 1.22-fold mass activity compared to the Pt NR/C electrode, and 1.14 and 1.51-fold compared to the commercial Pt/C electrode, respectively. Comparable durability is also established in fuel cells by using the accelerated degradation test, although Ag is included.


 Please wait while we load your content...
Please wait while we load your content...