Excellent electronic conductivity, insolubility and rate characteristics of DAAP based on chemical bonding with carbon fiber felt
Abstract
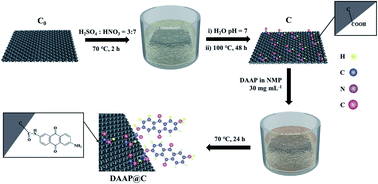
The use of certain small molecule aromatic carbonyl compounds (ACCs) as positive electrode materials in lithium ion batteries is a dilemma because of their good electrochemical properties (a high capacity density and good reversibility of the electrochemical reaction) and their fatal problems (a low electron conductivity and high solubility in organic electrolytes). To promote commercial application of ACCs, the synthesis and electrochemical behavior of an organic combination of carbon fiber felt and 2,6-diaminoanthraquinone (DAAP) were explored. The surface of carbon fiber felt after acidification (C) was rich with carboxyl groups, which reacted with the amine groups in DAAP molecules to form stable chemical bonds via amide bonds (–CO–NH–). The organic combination of carbon fiber felt and DAAP (DAAP@C) not only effectively enhanced the electronic conductivity and insolubility of the composites but also significantly improved their electrochemical performance (rate performance, cycle life, etc.). Compared with DAAP, the electronic conductivity, charge transfer resistance, lithium ion diffusion coefficient, initial capacity density (0.5C), and capacity retention rate (5C after 400 cycles) of DAAP@C were 111.1 S cm−1 (8.339 S cm−1), 103 Ω (418 Ω), 1.37 × 10−12 cm−2 s−1 (2.87 × 10−13 cm−2 s−1), 285 mA h g−1 (209 mA h g−1), and 80% (0%), respectively. This method, to graft soluble organic molecules onto tangible materials with high electronic conductivity, is an effective approach to simultaneously solve the problems of solubility and electronic conductivity inherent to most of small organic molecules with electrochemical activity, and with its use, the spring of organic electrode materials will come.


 Please wait while we load your content...
Please wait while we load your content...