Enhanced photochemical CO2 reduction in the gas phase by graphdiyne†
Abstract
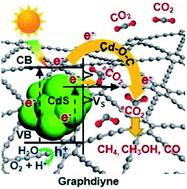
Photocatalytic CO2 reduction is promising for reducing the greenhouse effect and producing renewable energy, but still shows low activity and selectivity due to the ineffective utilization of photogenerated charge carriers and insufficient active sites for CO2 adsorption and activation. Taking CdS nanocrystals as a model semiconductor, we demonstrate that graphdiyne, a new type of two-dimensional carbon allotrope uniquely formed from sp- and sp2-hybridized carbon, enhances CO2 photoreduction over CdS with higher activity, selectivity, and stability in the gas phase without any sacrificial agent compared to graphene. Both experimental and theoretical results prove that the chemical bonding between graphdiyne and CdS and sufficient CO2 adsorption sites due to the strong interfacial interaction-induced sulfur vacancies in CdS and more electron-deficient acetylenic linkages in graphdiyne lead to more efficient electron transfer and storage for the subsequent CO2 reduction reaction. The excellent properties of graphdiyne make it promising for applications in solar energy conversion.


 Please wait while we load your content...
Please wait while we load your content...