Atomic-scale identification of influencing factors of sodium dendrite growth on different current collectors†
Abstract
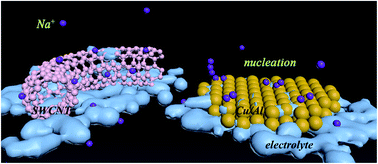
To design high-performance and safe Na metal anodes for rechargeable Na-metal batteries, the dendrite-growth mechanisms during the Na electroplating process need to be fully understood. Here, we provided a density functional theory (DFT) insight into essential mechanisms associated with Na dendrite formation on different current collectors, such as Cu, Al, and single wall carbon nanotubes (SWCNTs). We investigated the adsorption behavior of Na atoms on SWCNTs, Cu (111) surfaces, and Al (111) surfaces and further compared the stability of Na dimers. The “sodiophilic” properties and electronic configuration of Cu, Al, and SWCNTs were evaluated. Meanwhile, the electron transfer and the stability of Na dimers were estimated. For the adsorption of a single Na atom, the SWCNT, Cu and Al performed well with adsorption energies of −2.15, −2.93 and −2.24 eV, respectively. However, the Na dimer was not energetically favorable to form on SWCNTs. Based on Hirshfeld atomic charges and electron density distribution, the stable electron configuration of the SWCNT was found to play a critical role in dispersing Na adatoms. In addition, the vacancy defects in the SWCNT induced better “sodiophilic” properties and inhibited dendrite growth. Our results unraveled the possible mechanisms underlying dendritic electrodeposition of Na on SWCNTs, Cu and Al current collectors, indicating that SWCNTs can be a promising current collector to develop dendrite-free Na metal anodes for safe Na-metal batteries.
- This article is part of the themed collection: Journal of Materials Chemistry A HOT Papers


 Please wait while we load your content...
Please wait while we load your content...