Identification of active sites for CO2 hydrogenation in Fe catalysts by first-principles microkinetic modelling†
Abstract
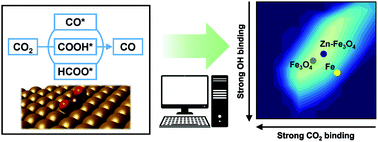
Active phase and reaction mechanism identification is imperative when designing catalysts for target reactions. Although computational analysis often suggests effective methods, the complexity and computational cost can be problematic, especially for Fe-based catalysts with various oxidation numbers. Herein, we elucidated the Fe phase involved in CO2 hydrogenation through a density-functional-theory-based energetics study and suggested promoters by performing microkinetic analysis. Our theoretical model suggested that (1) Fe3O4 is a major active phase for the reverse water–gas shift (RWGS) reaction; (2) direct dissociation of CO2 is preferred over hydrogenation; and (3) surface oxygen removal determines the overall reaction rate over most Fe surfaces. Using CO2 and OH binding energies as activity descriptors to screen promoters, Cu and Zn were found to accelerate the RWGS rate by facilitating OH removal. The proposed microkinetic modelling approach will aid in predicting complex heterogeneous catalysis processes involving diverse intermediates and surface phase transitions.


 Please wait while we load your content...
Please wait while we load your content...