Highly efficient removal of uranium from highly acidic media achieved using a phosphine oxide and amino functionalized superparamagnetic composite polymer adsorbent†
Abstract
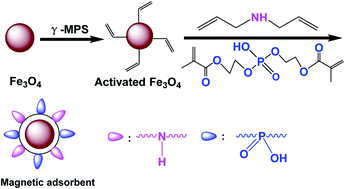
It is very important to develop magnetic adsorbents for the efficient extraction of uranium from highly acidic media. In this work, a novel magnetic adsorbent Fe3O4/P(DMAA–DMP) functionalized with phosphine oxide and amino groups has been synthesized by distillation–precipitation polymerization of diallylamine (DMAA) and di(methacryloxyethyl)phosphate (DMP). Surprisingly, Fe3O4/P(DMAA–DMP) not only showed an outstanding adsorption efficiency for uranium in highly acidic solution, but also had excellent acid resistance. The maximum adsorption capacity (qmax) of the magnetic adsorbent based on the Langmuir model could reach 196.4 mg g−1 even at a pH value of 0.5, which was much higher than that of all the magnetic adsorbents reported previously. And the structure and properties of the adsorbent did not change obviously after being immersed in HNO3 solution at a pH value of 0.5 for 24 h. Besides, the magnetic adsorbent also had a good selectivity for uranium and an excellent recyclability. XPS analysis confirmed that the outstanding adsorption capacity of the magnetic adsorbent for uranium was attributed to the strong complexation with phosphine oxide and amino groups. This work displays a simple route for developing a magnetic adsorbent for the highly efficient adsorption of uranium in highly acidic media, which is very significant for the recovery of uranium from highly acidic solution.


 Please wait while we load your content...
Please wait while we load your content...