Anti-freezing flexible aqueous Zn–MnO2 batteries working at −35 °C enabled by a borax-crosslinked polyvinyl alcohol/glycerol gel electrolyte†
Abstract
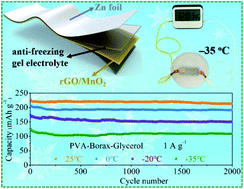
Flexible aqueous zinc-ion batteries (AZIBs) are promising to satisfy the emerging wearable electronics. However, conventional hydrogel electrolytes are unable to work at subzero temperatures because they inevitably freeze. In this work, a borax-crosslinked polyvinyl alcohol (PVA)/glycerol gel electrolyte is developed, in which glycerol can strongly interact with PVA chains, thus effectively prohibiting the formation of ice crystals within the whole gel network. Thanks to this, the freezing point of this gel electrolyte is below −60 °C, which allows it to work in extremely cold environments. Even at −35 °C, it still exhibits a high ionic conductivity of 10.1 mS cm−1 and great mechanical properties. On the basis of this anti-freezing gel electrolyte, a flexible quasi-solid-state aqueous Zn–MnO2 battery is assembled and realizes an impressive energy density of 46.8 mW h cm−3 (1330 μW h cm−2) at a power density of 96 mW cm−3 (2.7 mW cm−2) at 25 °C, outperforming nearly all the reported AZIBs. More importantly, when the temperature is reduced to −35 °C, a rather high energy density (25.8 mW h cm−3, 732 μW h cm−2) can still be achieved, and 53.3% of that value can be retained when the power density is increased to about 10-fold. This battery also shows excellent cycling durability (around 90% capacity retention over 2000 cycles) and great tolerance to various extreme conditions even when the temperature is down to −35 °C. These findings provide valuable insights into designing aqueous batteries/supercapacitors that can work in cold climates and high-altitude areas.
- This article is part of the themed collection: 2020 Journal of Materials Chemistry A most popular articles


 Please wait while we load your content...
Please wait while we load your content...