Hollow PtCu nanoparticles encapsulated into a carbon shell via mild annealing of Cu metal–organic frameworks†
Abstract
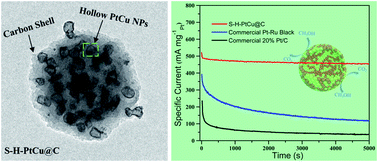
Alloying Pt with less expensive 3d transition metals to form bimetallic nanoparticles (NPs) has been proven to be an ideal strategy for the synthesis of catalysts, especially in the field of electrocatalysis. Unfortunately, these low-price 3d transition metals are easily leached from the alloy, thereby resulting in insufficient catalyst durability. To overcome this challenge as well as enhance the activity and durability of the alloy catalyst, spherical hollow PtCu nanoparticles (NPs) are encapsulated into a carbon shell via mild annealing of Cu metal–organic frameworks (S-H-PtxCuy@C). The carbon shell can effectively prevent the degradation and thus stabilize the PtCu alloy, and the hollow structure of PtCu NPs might enhance the activity. Consequently, the as-prepared S-H-PtxCuy@C catalysts exhibit excellent electrocatalytic performance toward the methanol oxidation reaction (MOR). Impressively, the S-H-PtCu@C catalyst can retain over 84% (444.3 mA mg−1) of its initial activity after 20 000 s durability measurement. Theoretical calculations reveal that the electron transfer between Cu and Pt results in electron redistribution, thereby weakening the binding strength of the adsorbate (e.g. COads) on Pt atoms and thus enhancing the MOR activity. The significance of this work is that it not only provides valuable information for the future design of highly robust alloy catalysts, but also sheds light on the development of MOF-derived functional nanomaterials.


 Please wait while we load your content...
Please wait while we load your content...