Catalyzing zinc-ion intercalation in hydrated vanadates for aqueous zinc-ion batteries†
Abstract
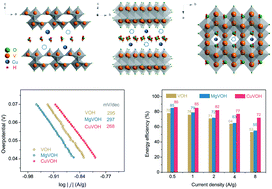
Hydrated vanadium pentoxide (VOH) can deliver a gravimetric capacity as high as 400 mA h g−1 owing to the variable valence states of the V cation from 5+ to 3+ in an aqueous zinc ion battery. The incorporation of divalent transition metal cations has been demonstrated to overcome the structural instability, sluggish kinetics, fast capacity degradation, and serious polarization. The current study reveals that the catalytic effects of transition metal cations are probably the key to the significantly improved electrochemical properties and battery performance because of the higher covalent character of 55% in the Cu–O bond in comparison with 32% in the Mg–O bond in the respective samples. Cu(II) pre-inserted VOH (CuVOH) possesses a significantly enhanced intercalation storage capacity, an increased discharge voltage, great transport properties, and reduced polarization, while both VOH and Mg(II) pre-inserted VOH (MgVOH) demonstrate similar electrochemical properties and performances, indicating that the incorporation of Mg cations has little or no impact. For example, CuVOH has a redox voltage gap of 0.02 V, much smaller than 0.25 V for VOH and 0.27 V for MgVOH. CuVOH shows an enhanced exchange current density of 0.23 A g−1, compared to 0.20 A g−1 for VOH and 0.19 A g−1 for MgVOH. CuVOH delivers a zinc ion storage capacity of 379 mA h g−1, higher than 349 mA h g−1 for MgVOH and 337 mA h g−1 for VOH at 0.5 A g−1. CuVOH shows an energy efficiency of 72%, superior to 53% for VOH and 55% for MgVOH. All of the results suggest that pre-inserted Cu(II) cations played a critical role in catalyzing the zinc ion intercalation reaction, while the Mg(II) cations did not exert a detectable catalytic effect.


 Please wait while we load your content...
Please wait while we load your content...