Synergism of surface strain and interfacial polarization on Pd@Au core–shell cocatalysts for highly efficient photocatalytic CO2 reduction over TiO2†
Abstract
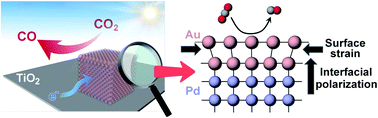
Solar driven conversion of CO2 into carbonaceous chemicals is a promising strategy to mitigate greenhouse gas emission and simultaneously store renewable energy. The rational construction of heterostructured cocatalysts represents an efficient method to improve the photocatalytic activity and selectivity for CO2 reduction. In this work, with Pd@Au core–shell nanostructures with tunable Au thickness as model cocatalysts, we demonstrate the synergism of surface strain and interfacial polarization for enhanced photoreduction of CO2 to CO. According to our experimental analysis and theoretical simulation, resulting from the mismatch in lattice parameters between the Pd core and the Au shell, compressive strain on the Au surface elevates the d-band center and improves the adsorption of the key intermediate *COOH. Meanwhile, charge polarization, driven by the difference in electronegativity between Pd and Au, accelerates the interfacial charge transfer and increases the electron density on the Au surface. It is found that both effects are dependent on the thickness of the Au shell. As a result, a three-atom-thick Au shell dramatically boosts the overall efficiency in CO2-to-CO conversion with an impressive activity of 166.3 μmol gcat−1 h−1 and selectivity of 90.6%. This study can be viewed as a means of designing photocatalysts via the simultaneous control of surface catalytic reactivity and interfacial charge transfer in cocatalysts.


 Please wait while we load your content...
Please wait while we load your content...