Plasmonically driven photocatalytic hydrogen evolution activity of a Pt-functionalized Au@CeO2 core–shell catalyst under visible light†
Abstract
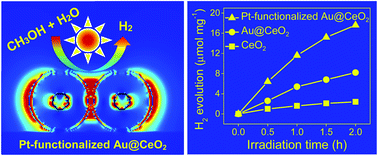
A ternary hybrid photocatalyst consisting of Au@CeO2 core–shell nanostructures coated with ultralow Pt-functionalized surface contents (Au@CeO2–Pt) was synthesized using a facile hydrothermal method. The Pt-decorated Au@CeO2 core–shell photocatalyst had a large Brunauer–Emmett–Teller (BET) surface area (86.50 m2 g−1) with an abundance of catalytically active sites. The hydrogen production rate over Au@CeO2–Pt under visible light (8.7 μmol mg−1 h−1) was much higher than that observed over binary Au@CeO2 (3.9 μmol mg−1 h−1) and pure CeO2 (0.9 μmol mg−1 h−1) catalysts. Enhancement of the hydrogen evolution reaction (HER) was attributed mainly to the surface plasmon resonance (SPR) effect of the Au cores, which was highly advantageous for light absorption and conversion. In addition, the Pt co-catalyst served as an electron sink that accepted photogenerated electrons migrating from the Au@CeO2 core–shells. This extended the lifetime of the photogenerated charge carriers and resulted in HER activity that was superior to that of the Pt-free photocatalysts. The large BET surface area of the ternary core–shell nanostructure and the presence of many catalytically active sites also enhanced the HER performance.
- This article is part of the themed collection: Editor’s Choice: Hydrothermal Materials Synthesis


 Please wait while we load your content...
Please wait while we load your content...