In situ construction of a MOF-derived carbon-encapsulated LiCoO2 heterostructure as a superior cathode for elevated-voltage lithium storage: from experimental to theoretical study†
Abstract
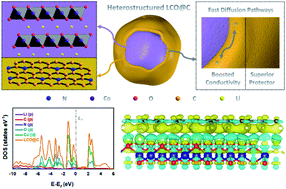
Lithium cobalt oxide (LiCoO2) is a promising cathode material for lithium ion batteries (LIBs). LiCoO2 achieves merely half of its theoretical specific capacity for commercial applications. Its intrinsic issues lead to significant structural instability and severe degradation of electrochemical performance. Herein, a strategy for a surface-modified LiCoO2 heterostructure by in situ metal–organic framework (MOF)-derived carbon-coating is proposed. The carbon-coated layer can not only reduce direct contact between the LiCoO2 bulk and electrolyte, but boost the electrochemical conductivity and strain structural distortion during lithiation/delithiation, further facilitating prolonged cyclability and distinguished rate performance. Moreover, the annealing temperature effect of fabricating such LCO@C electrodes was investigated systematically. N-doped LCO@C-700 delivered prolonged cycling stability (gravimetric/areal capacity of 171.1 mA h g−1/4.2 mA h cm−2 at 1C after 200 cycles) and superior rate capability (high capacity of 150.3 mA h g−1 even at a harsh current density of 10C) in the voltage range of 3.0–4.5 V. Density functional theory (DFT) calculations synchronously showed the electronic properties and Li-vacancy diffusion pathway of heterostructured N-doped LCO@C cathode material. These results indicate that electron-density charges, boosted electronic conductivity, and low energy barriers contributed to much faster lithium-ion storage kinetics of LCO@C particles than those of sole LCO. Our proposed in situ MOF-derived carbon-encapsulated strategy for cathode materials provides an innovative perspective and avenue for design of MOF-derived surface-modified heterostructure materials for LIBs.


 Please wait while we load your content...
Please wait while we load your content...