Revealing the failure mechanism of transition-metal chalcogenides towards the copper current collector in secondary batteries†
Abstract
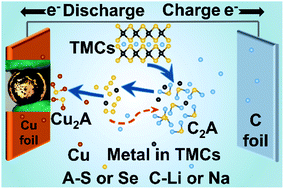
Transition-metal chalcogenides (TMCs) are attractive high-capacity anode candidates for secondary batteries, but they suffer from significant challenges including dramatic volume expansion and rapid capacity fading. Despite considerable progress, it was still a big challenge to achieve hundreds of stable cycles with high capacity retention. Herein, we reveal that the copper (Cu) current collector corrosion due to the sulphur (S) species attack leads to serious decay in anode performance in TMC-based batteries, as evidenced by the side-reaction product Cu2S generated via the consumption of S and Cu on the cycled TMC electrode. The electrochemical test and DFT calculations both reveal that the resultant Cu2S increases the resistance at interfaces and reduces the charge transfer, leading to a low capacity and short lifespan. By simply introducing a conductive layer between Cu foil and TMCs, the corrosive influence of Cu is remarkably suppressed, resulting in much enhanced cycling performance. At current densities of 0.5C and 1C, the Cu/2hTi/MoS2 sample delivered a high discharge capacity of 1010 and 740 mA h g−1 after 400 cycles, respectively. This work provides new insights to understand the fundamental mechanism of the electrochemical reactions and strategy to improve the energy storage ability of TMCs.


 Please wait while we load your content...
Please wait while we load your content...