Electrochemically controlled energy release from a norbornadiene-based solar thermal fuel: increasing the reversibility to 99.8% using HOPG as the electrode material†
Abstract
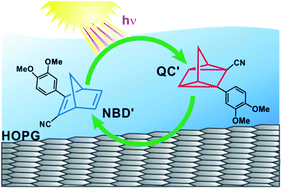
Solar energy conversion using molecular photoswitches holds great potential to store energy from sunlight in the form of chemical energy in a process that can be easily implemented in a direct solar energy storage device. In this context, we investigated the electrochemically triggered energy release of a solar thermal fuel based on the norbornadiene (NBD)/quadricyclane (QC) couple by photoelectrochemical IR reflection absorption spectroscopy (PEC-IRRAS). We studied the photo-induced conversion of the energy-lean 2-cyano-3-(3,4-dimethoxyphenyl)-norbornadiene (NBD′) to the energy-rich 2-cyano-3-(3,4-dimethoxyphenyl)-quadricyclane (QC′) and the electrochemically triggered reconversion using highly oriented pyrolytic graphite (HOPG) as an electrode material. We compared our results with the results obtained previously using Pt(111) electrodes and we characterized the photochemical and electrochemical properties of the storage system. NBD′ can be photochemically converted and electrochemically reconverted with very high selectivity. HOPG largely suppresses the unwanted catalytic reconversion which was observed on Pt(111). We performed repetitive cycling experiments for 1000 cycles to determine the reversibility of the system. Our results show that it is possible to reach reversibility above 99.8% using HOPG as an electrode material.
- This article is part of the themed collection: Journal of Materials Chemistry A Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...