Restraining the polarization increase of Ni-rich and low-Co cathodes upon cycling by Al-doping†
Abstract
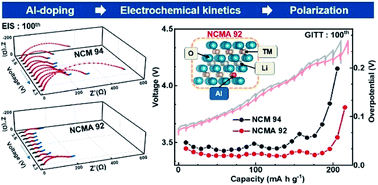
Ni-rich and low-Co cathodes (LiNixCoyMn1−x−yO2, x > 0.9, y ≤ 0.03) have the advantages of high capacity and low cost. However, the sharp increase in polarization upon cycling is unfavorable to the cycling performance. Herein, the mechanism of polarization increase is explored in detail, and Al-doping is proposed to restrain the polarization increase upon cycling. First, the electrochemistry test, in situ XRD, GITT and EIS are combined to provide new insights for understanding the polarization increase process. In addition, the mechanism of Al-doping which suppresses the polarization increase is also investigated. During cycling, the main cause for the increased polarization of Al-free LiNi0.94Co0.03Mn0.03O2 (NCM 94) cathodes is attributed to kinetic reasons. It is worth noting that the change of Li dynamics and charge-transfer impedance is more obvious at high SOC, which is consistent with the evolution of polarization. In contrast, because of the stabilizing effect of Al-doping on the structure, Al-doped LiNi0.92Co0.03Mn0.03Al0.02O2 (NCMA 92) cathodes can alleviate polarization increase during prolonged cycling. In detail, Al-doping reduces mechanical strain and relieves the accumulation of local internal stress at high SOC, which is conducive to improving the structural stability, maintaining the connection between primary particles, reducing the surface degradation along microcracks, and thus suppressing the deterioration of the electrochemical kinetics during cycling. Benefiting from the alleviative polarization increase, the NCMA 92 cathodes demonstrate a capacity retention of 92% after 100 cycles, while the NCM 94 only 69%. In this article, the insights on the polarization increase process of Ni-rich and low-Co cathodes will further help in the understanding of their cycling behavior and contribute to the efforts of improving their electrochemical performance.


 Please wait while we load your content...
Please wait while we load your content...