A bimetallic-MOF catalyst for efficient CO2 photoreduction from simulated flue gas to value-added formate†
Abstract
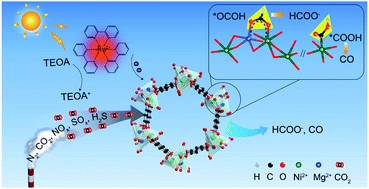
Direct CO2 conversion from flue gas into high-value products is of great significance not only in relieving environmental burden but alleviating the energy crisis by a low-cost and energy-saving avenue, yet few studies in this aspect have been reported. Herein, we report metal-node-dependent catalytic performance for solar-energy-powered CO2 reduction to formate in simulated flue gas by bimetallic Ni/Mg-MOF-74. The yield of HCOO− with Ni0.75Mg0.25-MOF-74 as a catalyst in pure CO2 is 0.64 mmol h−1 gMOF−1 which is higher than that of Ni-MOF-74 (0.29 mmol h−1 gMOF−1) and Ni0.87Mg0.13-MOF-74 (0.54 mmol h−1 gMOF−1), whereas monometallic Mg-MOF-74 is almost inactive, indicating that reactivity relies on metal nodes. In simulated flue gas without water vapor at 20 °C, ∼80% of the reactivity in pure CO2 is retained, with HCOO− generation reaching 0.52 mmol h−1 gMOF−1. This activity is comparable to that of the best MOF catalysts in pure CO2, demonstrating that Ni/Mg-MOF-74 not only overcomes the limitation from CO2 concentration, but also has good resistance to other gas components in flue gas at 20 °C. DFT calculations reveal the high output for HCOO− from two crucial factors: strong CO2 binding affinity of Mg sites, and the synergistic effect of Mg and Ni leading to the stabilization of the key *OCOH intermediate with an appropriate energy barrier. This work paves a new route for double-metal MOFs to enhance the CO2 photoreduction reactivity in flue gas.


 Please wait while we load your content...
Please wait while we load your content...