Insights into the chemical and electronic interface evolution of Li4Ti5O12 cycled in Li2S–P2S5 enabled by operando X-ray photoelectron spectroscopy†
Abstract
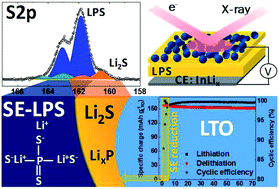
The (electro-)chemical reactivity and electronic properties across electrified solid–solid interfaces critically determine the functionality of solid-state electrochemical devices. To date, questions still arise in the scientific community due to the limitations of surface-sensitive characterization techniques, especially in post-mortem mode. Particularly, parameters such as the electrochemical stability of the solid electrolyte (SE) as well as the ionic and electronic percolation within the composite electrode are essential in order to push all-solid-state lithium-ion batteries (SSBs) towards practical applications. In this work, we highlight operando X-ray photoelectron spectroscopy (XPS) as a straightforward method to monitor the SE reduction within a composite electrode consisting of Li4Ti5O12 (LTO) as the electrode active material, (Li2S)3–P2S5 (LPS) as the SE and vapor-grown carbon fiber as the conductive additive under real working conditions. The results clarify previous discrepancies between experimental and theoretical results regarding the anodic stability limit of LPS and also reveal the limitations of post-mortem XPS measurements that can potentially lead to misinterpretations. We identify the reduction process of LPS which starts at 1.7 V vs. Li+/Li (1.1 V vs. InLix), resolved as a two-step process accompanied by the formation of electronically insulating Li2S and electronically conductive LixP species. Furthermore, the operando methodology resolves the LTO redox activity and the distribution of conductive and non-conductive LTO species as a function of their lithiation states. This allows a deeper understanding of both the lithium-ion transport through the composite electrode and the characteristic metal–insulator transition associated with LTO. Finally, we discuss how the interfacial reactivity between LPS and LTO impacts the electrochemical performance of SSBs.


 Please wait while we load your content...
Please wait while we load your content...